The beginnings of the KLS Martin Group date back to the year 1896 with the founding of our site in Mühlheim an der Donau in Germany. Today, the company consists of several production and sales companies worldwide, which were merged in 2004 under the umbrella brand KLS Martin Group.


In 1896 Ludwig Leibinger founded a watchmaking business and named it after his son Karl Leibinger. Very soon, he focused on the manufacture of surgical instruments.

Ludwig Leibinger hands over the business to his son Karl Leibinger.
On September 1st, 1923, Gebrüder Martin was founded for the production and sale of surgical instruments and all kind of metal products, as well as for the trading of own and foreign products. The fact that success at this time of dizzying inflation was only to be achieved in exports was also recognized by the then shareholders:
Soon, the associates Gotthilf and Wilhelm Martin left the company. Then, Fritz Martin took over the shares of his brothers. Managing Director of Gebrüder Martin was Rudolf Buck, who also took the initiative to merge other Tuttlingen-based companies in the medical technology sector and to transform them from competition into a cooperation. The vision that every shareholder of the group takes over part of the production program took shape.
The following legally independent production companies joined forces to sell their products by Gebrüder Martin:
Soon after the merger, Fritz Martin sold his shares to the other shareholders. The company name Gebrüder Martin, which is now well-known in international markets, is being continued.
The Second World War initially stopped the further development. A company building was completely destroyed in 1945 and the situation forced the employees to work in emergency quarters.
In the 1950s, new catalogs were published, and new product lines were also included in the program. Through its participation in international trade fairs and through worldwide marketing activities, the company established itself internationally.

The Karl Leibinger Medizintechnik company is then run by Ludwig Leibinger after the death of his father. Ludwig Leibinger expands the production program and the production facilities of the company.

After the sudden death of his father Ludwig Leibinger due to an accident in Mexico, Karl Leibinger takes over running the company at the age of 24. Under his leadership, further production facilities were built and new products and production technologies were introduced.

A new sales building was built in Ludwigstaler Str. 132 in Tuttlingen where Gebrüder Martin moved into in 1981.
Stuckenbrock Medizintechnik GmbH was founded.
Gebrüder Berchtold company left the group. The company shares were transferred to Trumpf GmbH + Co. KG, Ditzingen, Germany, and Fritz Hüttinger Medizintechnik GmbH + Co. KG, Freiburg, Germany. The two companies were now additional shareholders of Gebrüder Martin.
Stuckenbrock Medizintechnik GmbH bought the shareholder company H. + H. Handte GmbH & Co. KG and took over their company shares in Gebrüder Martin.
The shareholder companies Karl Vögele Medizintechnik GmbH and Stuckenbrock Medizintechnik GmbH were merged.
The company shares of Ueth & Haug GmbH in Gebrüder Martin were purchased by Karl Leibinger Medizintechnik GmbH & Co. KG and merged with it.
Stuckenbrock Medizintechnik GmbH bought the company shares of Josef Heiß OHG in Gebrüder Martin.
The company shares of Trumpf GmbH + Co. KG of Gebrüder Martin were taken over by Karl Leibinger Medizintechnik GmbH & Co. KG and merged with it.
The company Fritz Hüttinger Medizintechnik GmbH + Co. KG was renamed to Trumpf Medizin Systeme GmbH + Co. KG. After this, the company shares were taken over by Karl Leibinger Medizintechnik GmbH & Co. KG and another change of name into KLS Martin GmbH + Co. KG was performed. Today, the company specializes in high-frequency and laser devices.
The name KLS Martin Group was introduced as a umbrella brand. The "KLS" in the name stands for "Karl Leibinger Surgical".

Christian Leibinger joins the company as managing director and takes over the company management in the fifth generation.
On the occasion of the 90th birthday of Gebrüder Martin, the traffic circle in the Ludwigstaler Straße in Tuttlingen was officially renamed KLS Martin Platz. Parallel to the naming, a sculpture by the artist Jörg Bach was unveiled in the center of the circle. The official address of Gebrüder Martin is now: KLS Martin Platz 1 (formerly Ludwigstaler Str. 132)

KLS Martin GmbH + Co. KG moved from Umkirch to a new company building in Freiburg.

On January 20th, 2016, the visitor center KLS Martin WORLD was opened in Tuttlingen.

On October 11th, 2019, the training center KLS Martin WORLD North America was opened in Jacksonville, Florida.

The KLS Martin Group has acquired a stake in its strategic partner CADS GmbH, which develops visionary software solutions for industry and can optimally support the company with CAD automation as well as additive and subtractive manufacturing.
On May 1, 2020, the Leibinger family takes over the major business operations of Stuckenbrock Medizintechnik GmbH and its shares in Gebrüder Martin. The products are fully integrated into Karl Leibinger Medizintechnik.
On March 31, 2022, the company Mondeal Medical Systems GmbH from Mühlheim an der Donau is integrated into the company Karl Leibinger Medizintechnik.
On May 31, 2022, the company Rudolf Buck GmbH from Mühlheim an der Donau is integrated into the company Karl Leibinger Medizintechnik.
On December 31, 2022, the company Lawton GmbH & Co. KG from Fridingen an der Donau is integrated into the company Karl Leibinger Medizintechnik.
As of November 01, 2023, the three remaining German companies of the KLS Martin Group - Karl Leibinger Medizintechnik from Mühlheim an der Donau, Gebrüder Martin from Tuttlingen and KLS Martin from Freiburg - merge into one company and since then operate under the name: KLS Martin SE & Co. KG

The first subsidiary Martin Italia S.r.l. based in Milan, Italy, started its operations.

KLS Martin LP was established in Jacksonville Florida to distribute specialty products in the United States. Today KLS Martin LP sells products for craniomaxillofacial surgery, plastic surgery and neurosurgery in North America.
Business contacts with Japan had existed since the 1950s. In 1995, Nippon Martin K.K. established a subsidiary in Tokyo. In the same year, the subsidiary Martin Nederland/Marned B.V. in Huizen near Amsterdam went to the start.
In the course of the further expansion, the new warehousing center in Tuttlingen was established.
A representative office in Moscow was opened.
A representative office was opened in Shanghai.
A representative office was opened in Dubai. In the UK, in the center of Reading, the subsidiary KLS Martin UK Ltd. was founded.
New subsidiaries were established in Malaysia (KLS Martin SE Asia Sdn Bhd.), Brazil (KLS Martin do Brasil Ltda.) and in the Sydney metropolitan area (KLS Martin Australia Pty Limited).
In India, the subsidiary KLS Martin India Pvt Ltd. was opened.
In Penang the first production facility was opened outside Germany (KLS Martin Malaysia Sdn Bhd.).
In Taiwan, the subsidiary KLS Martin Taiwan Ltd. was opened.
With the founding of KLS Martin Manufacturing, LLC in Jacksonville, the first production facility was built outside Germany for implants. The focus is on the production of patient-specific implants by means of additive manufacturing processes.
The representative office in Shanghai is being changed to a subsidiary of Gebrüder Martin – KLS Martin Medical (Shanghai) International Trading Co., Ltd.
Foundation of the IPS® planning office in San Sebastián, Spain.
The company was founded to provide local surgeons with an improved service and to give them a deeper knowledge of our patient-specific solutions.
Foundation of KLS Martin de México S.A. de C.V., based in Mexico City, to support local dealers in marketing and selling our products on the Latin American market.
Foundation of the IPS® planning office in Cardiff, Wales.
The company was established to provide improved service to local surgeons and to give them a deeper knowledge of our patient-specific solutions.
The foreign subsidiaries in Italy, Japan and the Netherlands are given new names:

Ludwig Leibinger founds our site in Mühlheim an der Donau - at that time under the name Karl Leibinger Medizintechnik - and starts manufacturing surgical instruments. To date, our Mühlheim site has developed into a manufacturer of implants, surgical instruments, sterilization containers and surgical lights.
The use of high-frequency electricity in medicine begins. In our role as a pioneer, at that time still known as Fritz Hüttinger, we develop one of the first electrosurgical units in cooperation with the University Hospital of Freiburg. Numerous innovations followed, making electrosurgery an essential cornerstone to this day.
Since the founding, surgical instruments have dominated the portfolio. State-of-the-art stainless steel products are continuously being added to the range, once again highlighting our pioneering role.
Together with Prof. Champy, the scientific pioneer from Strasbourg, we develop the use of miniplate osteosynthesis in maxillofacial surgery and establish it as the “gold standard”. An important step helping people suffering from traumas or malformations of the face and skull.
Instruments with tungsten carbide inserts are added to the product portfolio. They feature a service life substantially longer than inserts comprised of conventional instrument steel. To allow for easy differentiation, they are fitted with a gold-plated ring and marketed under the name of TC Gold.
We start manufacturing surgical and examination lights. Due to its outstanding photometric features, the first halogen light already went on to become an international bestseller.
New technologies enable modern surgical techniques. In addition to surgical instruments and electrosurgical equipment, laser units are used for non-contact cutting, ablation and hemostasis. The MY 30 is the most compact Nd:YAG laser of its time.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
Offering the most comprehensive modular range of proven distractors for oral and maxillofacial surgery, we are considered the market and technology leader worldwide. The good scientific results as well as the handling proven in everyday clinical practice are widely documented in numerous publications.
The MY 40 laser is the first laser unit specifically designed for pulmonary surgery. The optimized wavelength enables parenchyma and lung function-preserving surgery. Today, the Limax® diode-pumped Nd:YAG laser is considered the most successful special laser in pulmonary surgery.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
Together with the Fraunhofer Institute, we develop the microStop® – the world's first sterilization container with a reprocessable sterile barrier. It can be sterilized as often as required and, due to an innovative indicator and germ barrier, does not require consumables such as a seal or filter.

We yet again set the standard in oral and maxillofacial surgery. With LevelOne, the innovative storage system for implants, instruments and screws. It is not only clearly arranged and compact, but can also be configured and combined individually.
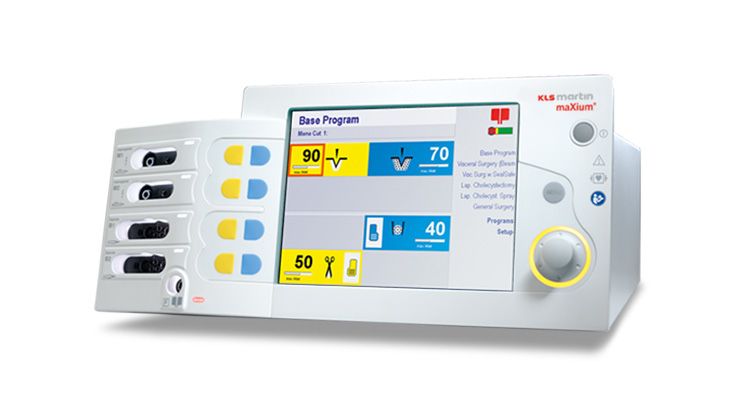
The maxium® electrosurgical unit enables intuitive operation of state-of-the-art high-frequency technology. A unique combination of user-friendly operation, outstanding design, performance and various special functions. In short: the optimal solution for all clinical disciplines.

Sealing technology enables new, more patient-friendly surgical methods: vessels can be cut and sealed in a single step and without ligation. With marSeal, we introduces the first reusable sealing instrument with integrated cutting function.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
SonicWeld Rx® sets new standards in osteosynthesis. The welding process supported by ultrasonic technology anchors the resorbable material deep inside the bone structure, resulting in three-dimensional primary stability on an unprecedented scale.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
Surgical lighting using LEDs to provide illumination are now state of the art. We are one of the first manufacturers to employ this innovative technology. The marLED® V series still impresses today with its design, which was awarded the Reddot Design Award and the Focus Green Silver.
Modelled on the knee joint replacement, the CapFlex PIP was developed as a surface replacement for the interphalangeal joint. The product offers decisive advantages such as large bony supporting surfaces for high primary stability.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
With its modular marWorld® concept, we offer individual and cross-product complete solutions for the operating room, all from a single source. This applies both to the construction and equipping with our modular OR system as well as to advisory support during the entire process - from conception to planning and implementation. The project business around marWorld® remained part of our offering until the end of 2021.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
With IPS Implants®, we design and manufacture precisely fitting individual implants based on patient data. Two years later, the IPS Gate®, with its standardized and digitized workflow, enables safe and convenient case handling and networks surgeons around the world.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
 By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
By loading the video, you accept YouTube's privacy policy. To play, accept YouTube cookies.
The intuitive IPS CaseDesigner® software enables the planning and simulation of orthognathic interventions based on individual patient data sets. Implementation in the OR is performed with splints or precisely fitting patient-specific implants.
Patient-specific solutions for the correction of complex malpositions of the upper extremities represent considerable relief for the treating surgeon. For example, this method allows sustained treatment of the Madelung deformity of the forearm for the very first time.