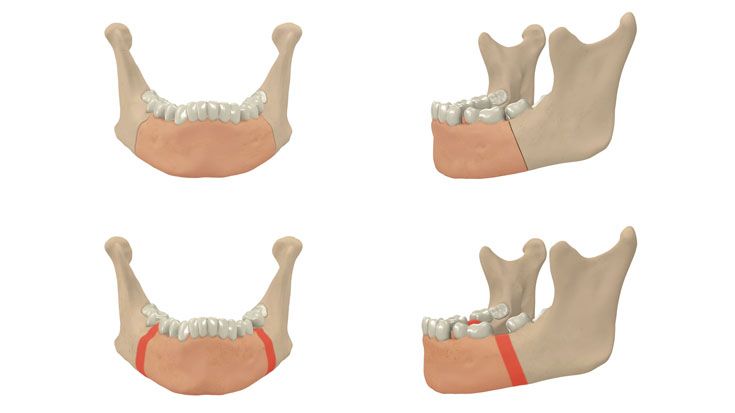
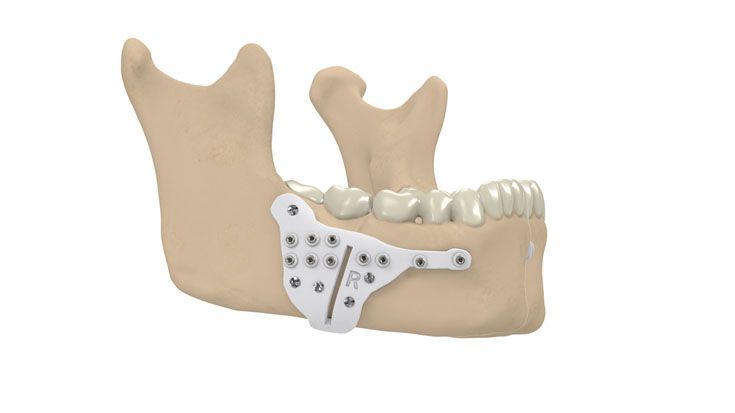
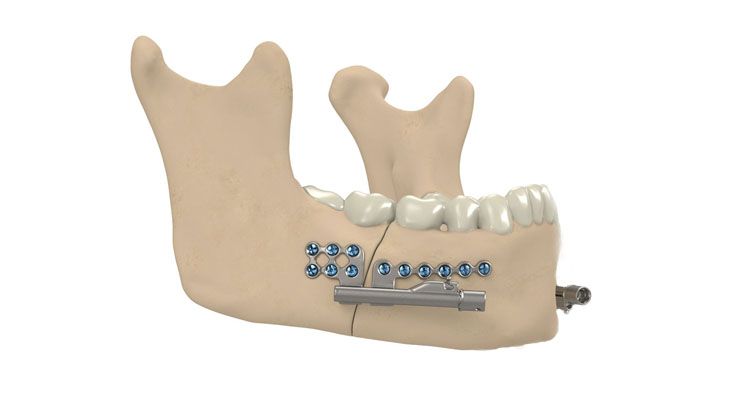
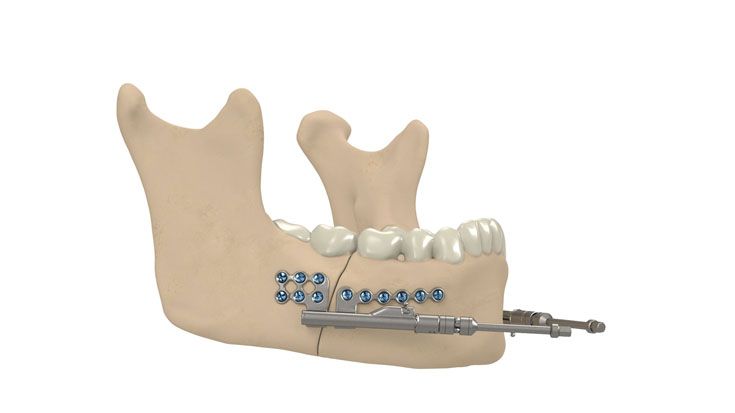
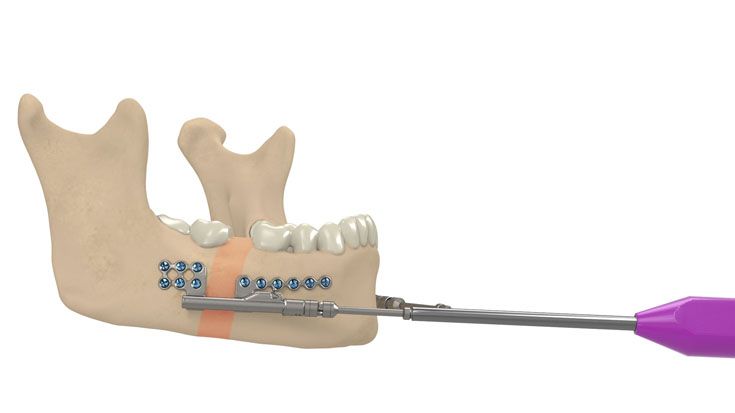
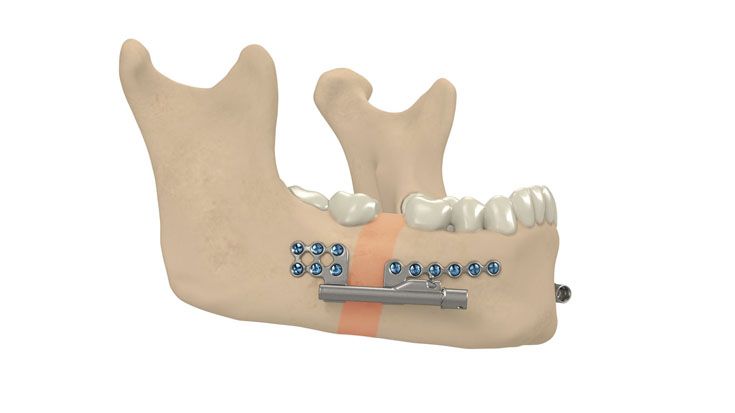
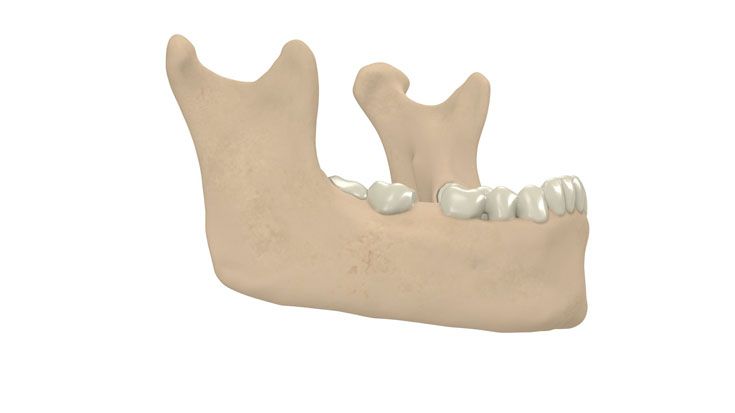
IPS Implants® Distraction
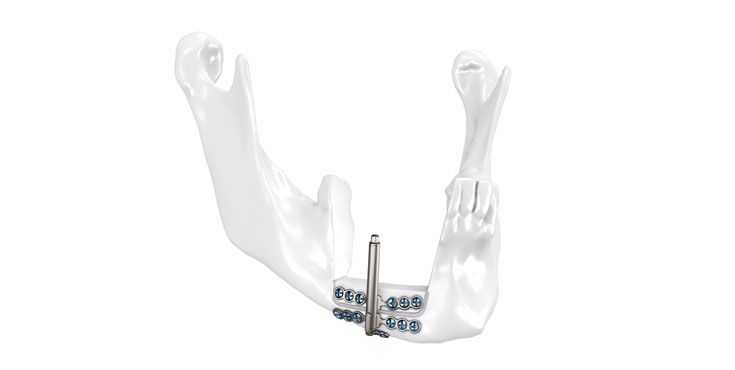
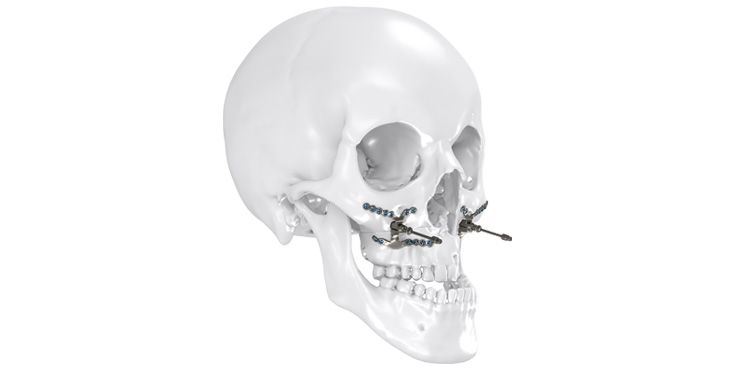
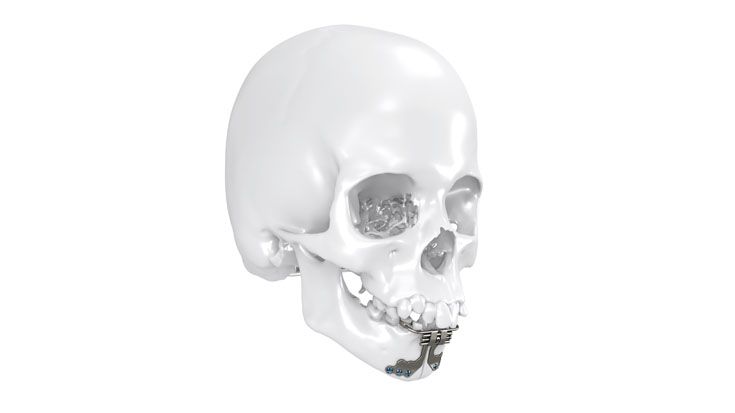
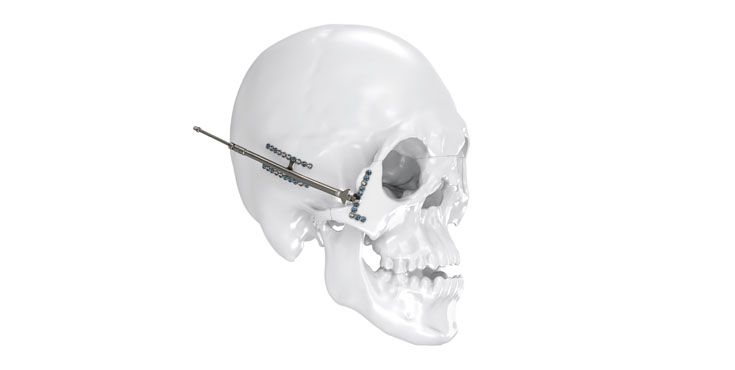
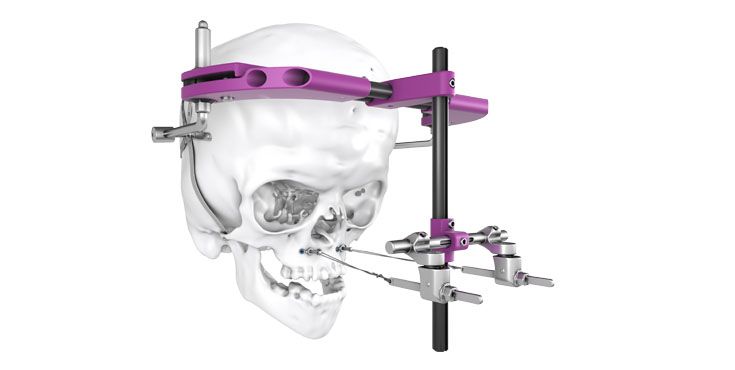
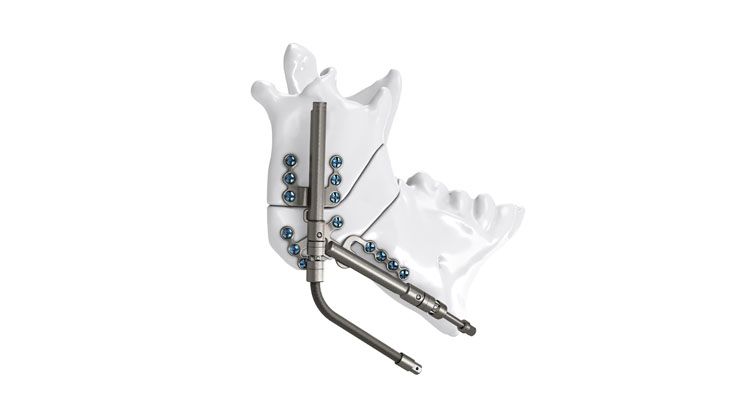
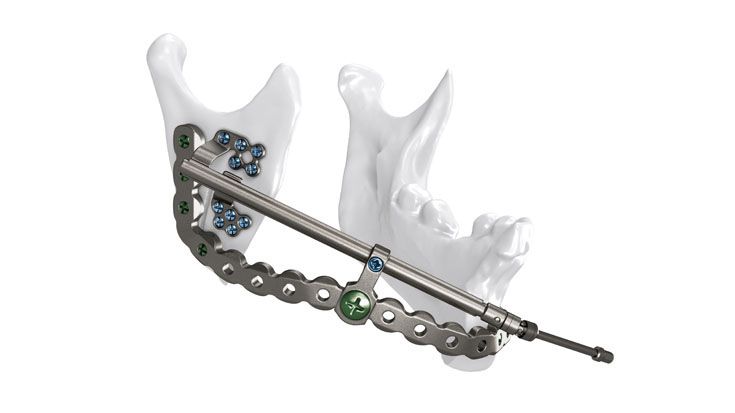
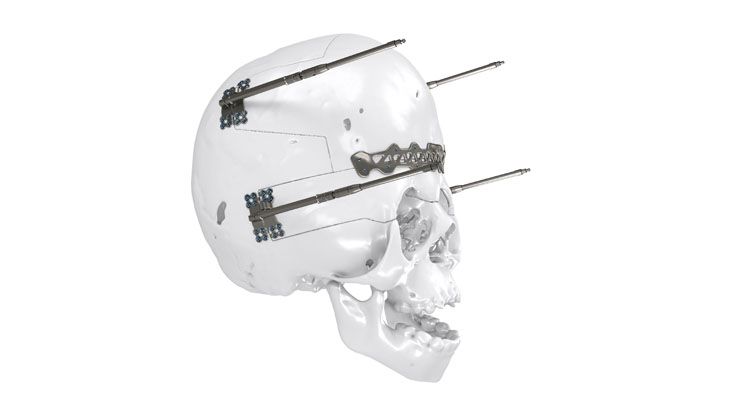
As the world market leader in the fields of distraction osteogenesis and patient-specific implant solutions, we have combined our expertise in both areas to set a new milestone in distraction techniques.
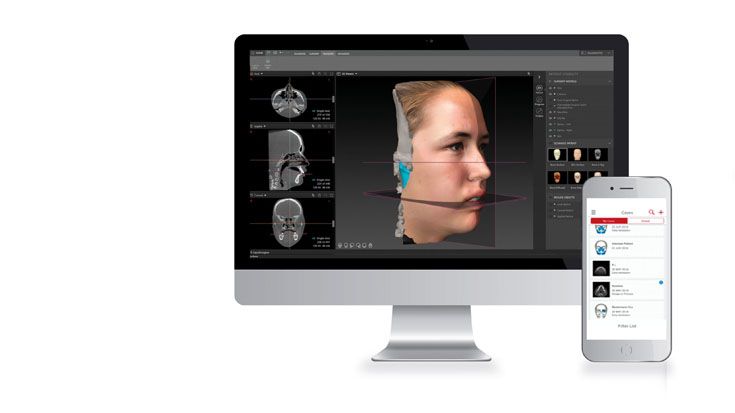
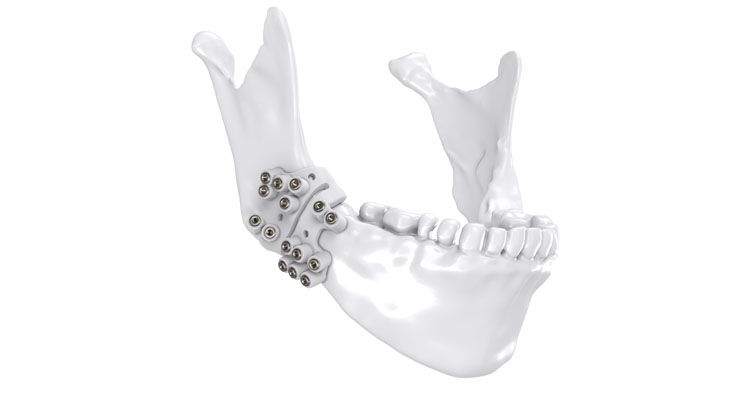
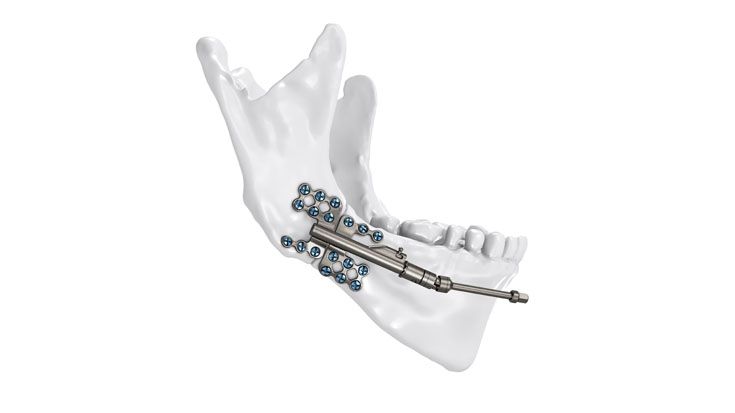
Based on our well-established distraction systems and the use of modern IPS® technologies we developed IPS Implants® Distraction, a new distraction solution, that is individually adapted to the patient. As part of the IPS® product family, IPS Implants® Distraction is supported by an easy and efficient process – from the planning up to the functionalized distractor.